Nov, 2023
Dhinakar Kompala gave an oral presentation at the International Forum on Cell Manufacturing & Engineering in Berlin, Germany on the use of Sudhin BioSettlers to recycle organoids, such as IPSC clusters, from the bottom of BioSettlers back to the bioreactors while removing the single cells and spent media from the top of BioSettler.

Oct, 2023
Dhinakar Kompala presented an invited 20 minute webinar, as a part of the Good Food Institute's monthly collaborative seminar series in October, discussing novel applications of Sudhin Biopharma's Inclined Settling Bioreactor for manufacturing cultured meat and inviting collaborators from this growing industry.
Dhinakar Kompala presented a poster at RAFT (Recent Advances in Fermentation Technology) conference on the successful application of BioSettlers to break oil-water emulsions generated during in situ product recovery in extractive fermentations and selectively remove the oil phase containing the toxic metabolite product. This poster followed an earlier podium presentation at the same conference by our BioMADE project collaborator Amyris's Chandresh Thakker on their successful use of Sudhin BioSettler (25 Liter) attached to their 300 Liter fermentor for in situ product extraction.

Sep, 2023
Dhinakar Kompala has been invited to give an oral presentation on “Inclined Settling Bioreactor for manufacturing of cell and gene therapy products” at the ECI conference Single Use Technologies VI in Boston in the “Emergent” technologies session on September 12th. Following the individual podium presentations in this session, we had an extended Question & Answer session and a panel discussion on Emergent Single Use Technologies on the same venue.

July, 2023
Sudhin Biopharma scientist demonstrate growth of CHO cells inside the newly developed Inclined Settling Bioreactor with single use disposable fluorescent dye sensors and thermocouple to measure the cell culture parameters of pH, D.O. and Temperature and automation and control system developed by Rockwell Automation to sparge controlled mix of air, oxygen and carbon dioxide to control the measure cell culture parameters.

April, 2023
Dhinakar Kompala presents an invited talk at the BioMADE member meeting in Minneapolis, discussing the novel applications of Sudhin BioSettlers to break oil-water emulsions generated during in situ product recovery in extractive fermentations and selectively remove the oil phase containing the toxic metabolite product.

Feb, 2023
Dhinakar Kompala presented an invited talk on “BioSettler technology and its evolution in to an Inclined Settling Bioreactor for growing cell and gene therapy products” to the Colorado Emerging Medical Device in-person meetup at the Broomfield offices of Aktiv Pharma Group.
Dec, 2022
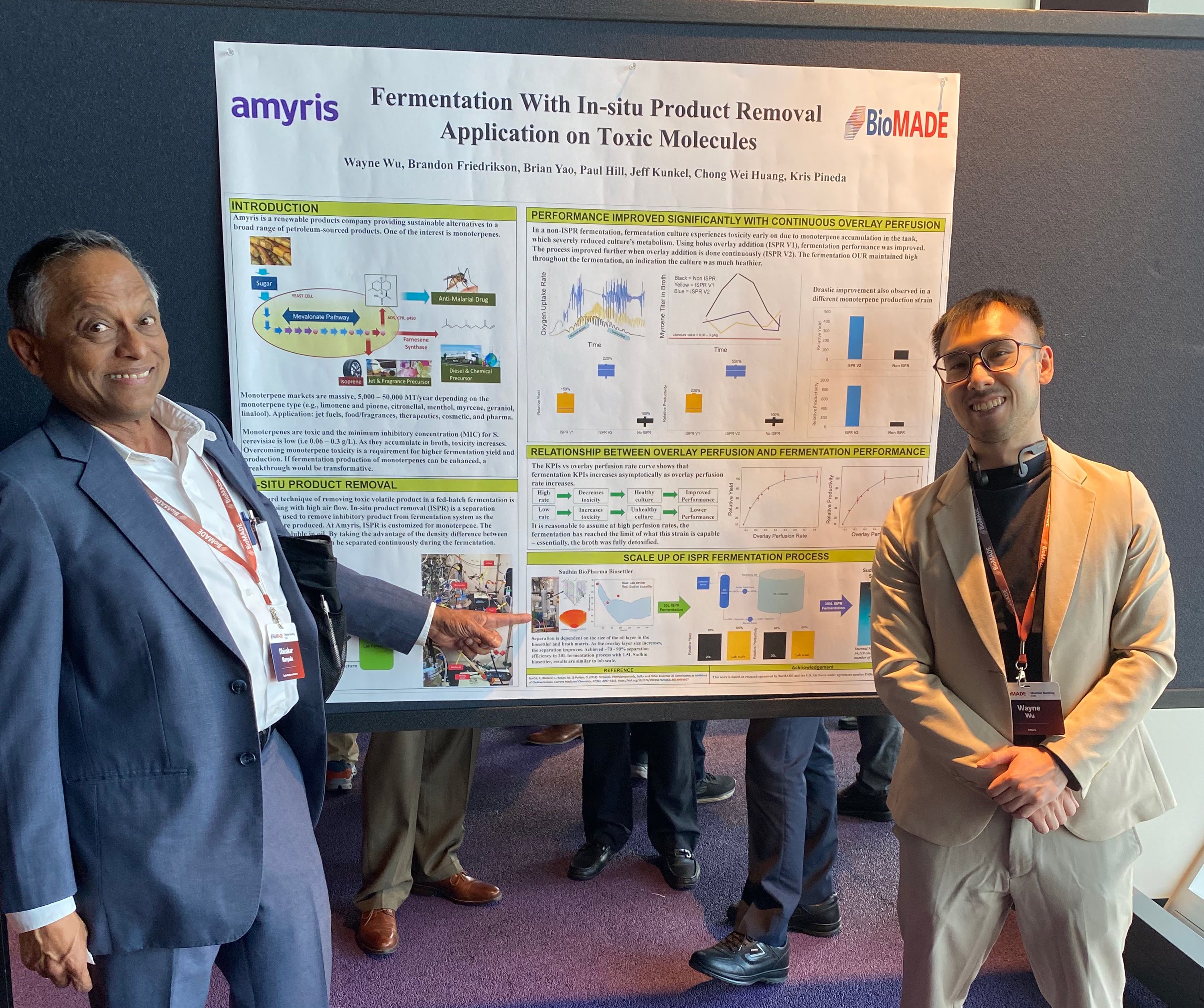
Sudhin BioSettler was demonstrated to be highly efficient at breaking oil-water emulsion and removing the oil layer continuously from the top, while the water phase is removed from the bottom of BioSettler. Based on these preliminary results reproduced at a synthetic biology company in California, Amyris, we were selected for a new R & D grant of $720,000 (Sudhin’s portion) from a new Manufacturing USA consortium, BioMADE with support from Schmidt Futures and started collaborating with Amyris after the final approval of this grant in April 2023.
Oct, 2022
Dhinakar Kompala presented a poster on "Integration of low pH viral inactivation and primary clarification of CHO cell culture broth inside Sudhin BioSettlers" at the ECI conference on Integrated Continuous Bioprocessing conference in Barcelona, Spain.
Sep, 2022
Dhinakar Kompala presented an invited talk on "Gentle concentration and harvest of IPSC clusters and organoids using Sudhin BioSettlers" at the Cell & Gene Therapy Manufacturing and Commercialization track of the BioProcess International / Biotech Week Boston conference.
July, 2022
Responding to a customer request, Sudhin scientists demonstrated the feasibility of breaking oil-water emulsion created by vigorous agitation by an impellor and pumped into the bottom two side ports of a 1 liter BioSettler at an inlet flow rate of about 7.5 liters/hour. The oil and water (colored red for easy visibility) phases are separated very quickly inside the BioSettler due to its numerous inclined surfaces and continuously removed from the top port and the bottom port respectively.

June, 2022
Dhinakar Kompala updated the funding agency Advanced Regenerative Manufacturing Institute (ARMI)’s BioFab USA, a DoD supported Manufacturing USA consortium and its fellow members attending the Annual Meeting in the Millyard at Manchester, New Hampshire of the progress to date on the project to develop an "Inclined Settling Bioreactor for Manufacturing Cell Therapy products" in a poster shown here.
March, 2022
Our smallest BioSettler75-250 has been demonstrated successfully by Pluristyx, Inc. a collaborator on our Advanced Regenerative Manufacturing Institute (ARMI) funded project to retain and recycle induced pluripotent stem cell (IPSC) clusters to an attached bioreactor, while removing the spent media containing smaller dead cells and cell debris via the top effluent port.
The final objective for this project is to develop a novel Inclined Settling Bioreactor (ISB) by installing single use disposable fluorescent dye sensors for pH and DO from another collaborator on this project, Scientific Bioprocessing, Inc. and controlling these key cell culture parameters inside the BioSettler and demonstrate mammalian cell culture inside this novel ISB.
October, 2021
Sudhin Biopharma is happy to announce the issuance of two US additional patents US 11,148,076 B2 and US 11,185,799 B2 (continuation on their two earlier patents) and issuance of growing number of foreign patent grants on an earlier US patent.
July, 2021
Braden Smith, Christopher Glascock and Dhinakar Kompala of Sudhin Biopharma present a short video highlighting the successful demonstration of "Sudhin BioSettlers for Clarification of Cell Culture Broth from Fed-batch Bioreactors" at the annual meeting (virtual and in-person) of the National Institute for Innovation in Manufacturing Biopharmaceuticals (NIIMBL) in Washington, D.C.
January, 2021
Dhinakar Kompala presents a talk on "Measuring pH and Dissolved Oxygen inside BioSettlers" at the Virtual Conference organized by Scientific Bioprocessing Inc. on "Oxygen and pH Sensing in Cell Culture: Latest Advances"
December, 2020
As the COVID year 2020 is coming to an end, We express our sympathies for many who have suffered from this pandemic. On a contrived positive note, We are happy to witness the rapid emergence of novel vaccines and antibody therapeutics, which are positive developments for the whole Biopharma industry.
At an individual company level, We are happy that my early stage companies, Sudhin Biopharma Co (US) and its subsidiary Sudhin Biotech Private Limited (India), have survived the "valley of death" during this pandemic year. In addition to the issuance of two US patents on our novel BioSettlers, early (but delayed) successful trials at select early adopter companies, we are excited to share the news of our selection for funding by Advanced Regenerative Manufacturing Institute (ARMI), a DoD funded Manufacturing USA institute.
The objectives of this new ARMI project are to demonstrate initially the use of BioSettlers for selective removal of spent media and concentrated harvest of stem cell clusters and to develop our BioSettlers over the next two years through the integration of single use fluorescent dye sensors for pH and DO into novel intelligent settling bioreactors for expansion of stem cells and concentration/harvest of stem cell clusters or organoids or viral vaccines or exosomes.
August, 2020
We have introduced the smaller BioSettler75, in response to the wishes of early testers of BioSettler150, for novel applications such as primary clarification, retention of stem cell clusters during media exchange, etc.

March, 2020
We are happy to announce the granting of first two U.S. patents from Sudhin Biopharma Company's larger patent portfolio on our BioSettler technology: U.S. Patent # 10576399 granted on March 3rd, 2020 and the U.S. Patent # 10596492 to be granted on March 24th, 2020.
October, 2019
Following the purchase of several BioSettler150 by a cell therapy company developing beta cell isles for transplantation into type I diabetics, Sudhin Biopharma joins a new Manufacturing USA consortium called "Advanced Regenerative Manufacturing Institute" ARMI/BioFab USA, sponsored by the US Department of Defense.
June, 2019
Clarification of CHO cell culture broth is successfully demonstrated inside our first BioSettler150 at the process development laboratory of a big biopharma company, after learning from the initial unsuccessful trial experiences at another big biopharma company in March 2019.
December, 2018
Dhinakar Kompala presented an oral presentation on “High Intensity production of a natural sweet protein in continuous perfusion cultures of recombinant yeast cells” at the Food Innovation and Engineering (FOODIE) Conference in Napa, CA during December 2 - 4, 2018.
Sudhin Biopharma Co. sells 3 of its single use disposable plastic settlers to a cell therapy company in Cambridge, MA for separation of β islet cells from induced pluripotent stem cells for type 1 diabetes cell therapy.
October, 2018
Dhinakar Kompala presented an invited talk on “Continuous harvesting of secreted products from a high cell density perfusion bioreactor of Pichia Pastoris (& CHO) cells“ in the session on Microbial Manufacturing at the BioProduction Congress in Dublin on October 9th, 2018.
Dhinakar presented an invited talk on “Single Use Disposable Cell Settler” at the symposium in honor of his Ph.D. mentor D. Ramkrishna’s 80th birthday symposium during the annual national meeting of the American Institute of Chemical Engineers in Pittsburg, PA during October 28 - 31, 2018.
September, 2018
Dhinakar Kompala presented an invited talk on “Single Use Disposable BioSettler” at the Manufacturing Summit of University of Massachusetts Lowell at the UMass Club in Boston on September 10th.
Dhinakar presented a poster presentation on the same topic “Single Use Disposable BioSettler” at the ECI conference on Single Use Technologies III in Snowbird, UT during September 23 - 26, 2018.
August 8th, 2018
Sudhin Biopharma sells its first 6” plastic settler to the University of Technology Sydney Australia for perfusion studies on algal cultures. This first sale follows the supply of several of these devices in July 2018 to a Tier 1 member of NIIMBL for their in-house testing of this device for clarifying cell culture broth as an alternative to traditional centrifuges.
May 22nd, 2018
Biomanufacturing Training & Education Center (BTEC) of North Carolina State University in Rayleigh, NC as the lead organization receives a grant from NIIMBL for a collaborative project with Sudhin Biopharma on a project titled “Development of a Gene Therapy Vector Production Platform”. BTEC will test the usage of Sudhin Biopharma’s single use disposable BioSettler in the continuous manufacturing of Adeno-Associated Virus (AAV) gene therapy vectors from insect (sf9) and Human Embryonic Kidney (HEK) cells.
May 17th, 2018
Dhinakar Kompala, CEO of Sudhin Biopharma Co. launches and presents its first product, a 6” diameter single use disposable BioSettler to the project lead at the collaborating Tier 1 member of NIIMBL at its second annual members only National Meeting. This event is featured in the website of NIIMBL with this news item (https://niimbl.force.com/s/news-list) and photograph:
First NIIMBL Project Output Revealed at National Meeting
Dr. Dhinakar Kompala of Sudhin Biopharma Co. unveils single-use cell settler.NIIMBL continues to build momentum toward new, innovative biomanufacturing technologies. That momentum was on display at the National Meeting as Dr. Dhinakar Kompala of Sudhin Biopharma Co., an early SME member, showed off the company’s new single-use compact settler for clarifying cell culture broth. The settler is among the first outputs of a NIIMBL-funded project.

May 16th, 2018
Dhinakar Kompala gave this 90 second talk showcasing the latest innovation of Sudhin Biopharma Company, at the 2nd annual meeting of National Institute for Innovation in Manufacturing Biopharmaceuticals (NIIMBL) in the auditorium of the National Academy of Sciences in Washington D.C.
March 2nd, 2018
Sudhin Biopharma receives a Quick Start Project award of $1.5 Millions from National Institute for Innovation in Manufacturing Biopharmaceuticals (NNIMBL), with in-kind cost-share commitment of another $0.5 millions from a Tier 1 industrial member of NIIMBL. The project, titled “Scale-up Studies on Single Use Settlers for Clarifying Cell Culture Broth,” aims to develop a disposable plastic device for clarifying cell culture broth from a single-use bioreactor. This process is currently done by centrifugation and/or using depth filters. However, removing cells through these methods becomes challenging at high cell densities and at large scale. Through this single-use disposable settler device, cells can be separated from the secreted protein product to produce a significantly clarified product harvest stream.
Sudhin Biopharma Co. will produce and deliver 6- inch, 12-inch and 24-inch diameter single use disposable plastic settlers as part of the project. These devices will also be demonstrated as selective cell retention devices to remove dead cells and cell debris through the settler’s top effluent and to return the live and productive cells to a continuous perfusion bioreactor, which can then be operated at high cell densities, viabilities and productivities over months-long culture duration.
Oct 17th, 2017
Dhinakar Kompala presents an invited talk on "Novel compact cell settler for high cell density perfusion bioreactor cultures of microbial (and mammalian) cells" at the BioProduction Congress in Convention Centre Dublin, Ireland.
https://lifesciences.knect365.com/bioproduction/speakers/dhinakar-kompala
Aug 2017
Sudhin Biopharma Co. publishes its first peer-reviewed article: Freeman, Cassandra A., Premsingh S.D. Samuel, and Dhinakar S. Kompala, "Compact cell settlers for perfusion cultures of microbial (and mammalian) cells", in a special issue of Biotechnology Progress 33:913-922 (2017). DOI 10.1002/btpr.2533
May 4th, 2017
Dhinakar Kompala presents a 2 minute introduction to Sudhin’s innovation at Small and Medium Enterprises Innovation Showcase during the Launch or Kickoff National Meeting of the newly funded National Institute for Innovation in Manufacturing Biopharmaceutics, of which Sudhin Biopharma just joined as a SME member.
Feb 15th, 2017
Second beta testing of our Stainless Steel Compact Cell Settler has commenced at a large biopharmaceutical company in Sweden. More devices are available for beta testing by other interested companies.
Dec 1st, 2016

Sudhin Biopharma Company has been awarded a National Science Foundation (NSF) Small Business Innovation Research (SBIR) Phase I grant for $225,000 to conduct research and development (R&D) work on demonstrating the power of our compact cell settler to achieve high cell densities and productivities in continuous perfusion bioreactor cultures of recombinant mammalian cells.
With this new grant funding, we plan to culture (i) recombinant Chinese hamster ovary cells and (ii) murine hybridoma cells in continuous perfusion bioreactors attached with our 4" or 6" compact cell settler and demonstrate that dead cells and cell debris can be removed selectively via the settler top effluent while the live and productive cells are recycled continuously to the bioreactor. Our patent-pending compact cell settler technology is a more efficiently scaled version (with much smaller footprint and higher settling capacity) of the proven "inclined settler" technology, which has been used successfully in commercial production of some therapeutic biologics. We expect to reproduce for the mammalian cell cultures our recently obtained excellent results for the smaller and hence more challenging yeast Pichia pastoris cells, demonstrating very high cell densities and productivities in continuous perfusion bioreactor for over two months without any membrane clogging or protein sieving challenges seen with other cell retention devices.
"The National Science Foundation supports small businesses with the most innovative, cutting-edge ideas that have the potential to become great commercial successes and make huge societal impacts," said Barry Johnson, Director of the NSF’s Division of Industrial Innovation and Partnerships. "We hope that this seed funding will spark solutions to some of the most important challenges of our time across all areas of science and technology."
Oct 15th, 2016
Beta testing of our Novel Compact Cell Settler has commenced at a large biopharmaceutical company in USA. More devices are available for beta testing by other interested companies.
Jun 8th, 2016
Dhinakar Kompala discusses his opinions about the advantages of continuous perfusion bioreactor culture over fed-batch bioreactor culture in a brief interview with a contract manufacturing company Patheon at the BIO International Convention in San Francisco, CA.
May 9th, 2016
Dhinakar presents the latest results from the novel compact cell settlers to achieve very high cell densities of yeast Pichia pastoris cells secreting a recombinant protein, pro-glargine, an insulin analog continuously harvested from a 5 liter perfusion bioreactor.
Feb 11th, 2016
Sudhin Biopharma Co. receives an Early Stage Capital and Retention grant of $50,000 from State of Colorado Office of Economic Development and International Trade. This is the first installment out of a total grant of $250,000 over three years, providing matching funds to other funding sources, including contracts, grants and investments.
Dec 19th, 2015
Dhinakar presents an invited talk on “Novel Compact Cell Settlers for Perfusion Cultures of (Microbial and Mammalian) cells” at the Bioprocessing India 2015 conference at Indian Institute of Technology, Madras. See below for the slides.
Also presented as a poster paper at the ECI conference on “Integrated Continuous Bioprocessing” in Berkeley, CA on Nov 3rd, 2015.
Dec 10th, 2015
Sudhin Biopharma Co. receives a Phase IB supplemental funding of $29,999 to the Small Business Innovative Research (SBIR) Phase I grant from the US National Science Foundation.
Sep 1st, 2015
Dr. Niles Utlaut, M.D., an angel investor, joins our newly constituted Board of Directors, along with our first angel investor, Dr. Sushila Kompala, M.D.
Mr. Khishore Pridhivi, MBA, joins the Board of Directors of our majority-owned subsidiary company in India, Sudhin Biotech Private Limited, along with Mr. Srinagesh Talatam, MBA.
Founder and CEO, Dr. Dhinakar Kompala, Ph.D. serves as the Chairman of both these Boards.
Jul 1st, 2015
Sudhin Biopharma Co. receives a Small Business Innovative Research (SBIR) Phase I grant of $150,000 from the US National Science Foundation.
Apr 21st, 2015
Dhinakar S Kompala presents at American Chemical Society's Entrepreneur Showcase West in San Diego, CA.